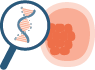
PGT-A Plus
PGT-A Plus
Our most advanced 4-in-1 genetic test offers increased accuracy and confidence for embryo transfer.

Combines two in-house validated technologies (NGS + SNPs analysis)

Superior accuracy and enhanced confidence to detect any abnormalities